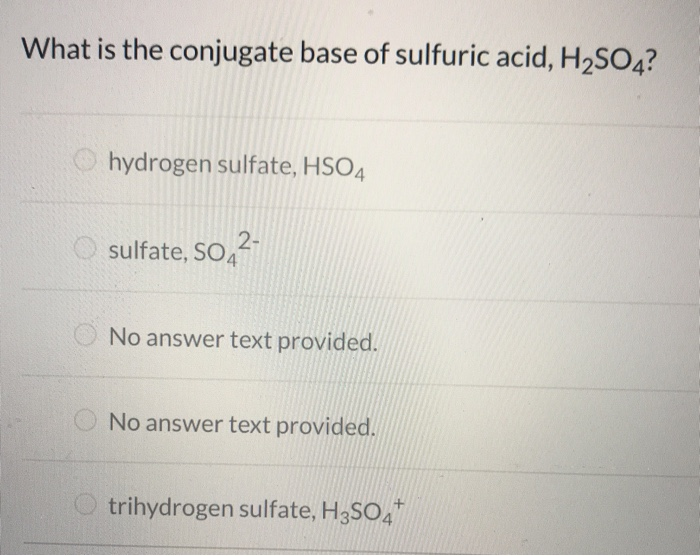
What is the conjugate base of h2so4
What is the conjugate of H 2 SO 4? A non-metallic element is converted into a compound X after a series of reactions. A little amount of X when tested with blue litmus turns to red. X on complete reaction with another compound Y gave the product which did not respond to litmus test.
Identify the acid, base, conjugate acid and conjugate base in the following reaction. HSO4" aq …. A: Acid Base chemistry. Q: Which statement is true of this chemical equation? Q: acid, base, conjugate acid, and conjugate base.
What is the conjugate base of h2so4
Wiki User. They are the products of an acid-base reaction by the Bronsted-Lowry definition. Conjugate base. HSO4 -. Nope, itsHSO H2SO4 is already a strong acid. If you mean what is the conjugate base, then the answer is HSO The conjugate base of a weak acid is always a strong base. You mean sulfuric acid. The conjugate base of HF is the fluoride ion F-.
If you mean what is the conjugate base, then the answer is HSO Keeping it similar to the general acid properties, Arrhenius acid also neutralizes bases and turns litmus paper into red. A strong acid will form a weak….
.
See this Socratic answer. A conjugate base contains one less H atom and one more - charge than the acid that formed it. Let us take the example of bicarbonate ions reacting with water to create carbonic acid and hydronium ions. It has one less H atom and one more — charge. Acid strength is determined by the amount of that acid that actually ionizes. All other acids are weak acids and ionize to a much lesser amount. All acids have a conjugate base.
What is the conjugate base of h2so4
What is the conjugate of H 2 SO 4? A non-metallic element is converted into a compound X after a series of reactions. A little amount of X when tested with blue litmus turns to red.
Elite turnaround specialists jobs
H2O… A: conjugate base is formed when an acid donates a proton. A: Interpretation- conjugate base. A: According to Bronsted-Lowry concept an acid is a proton donor and a base is a proton acceptor. What is the conjugate base for H2SeO3? A: The acid given is phosphoric acid i. Q: 1 Complete the following reactions and identify the acid, base, conjugate acid and conjugate base. HCO,- 3. Byju's Answer. Bronsted-Lowry base in inorganic chemistry is any chemical substance that can accept a proton from the other chemical substance it is reacting with. They are the products of an acid-base reaction by the Bronsted-Lowry definition. HF is an acid and F- is its conjugate base. A: Ans given as follows. Still have questions? H2O 3. The conjugate acid is H2CO3.
The magnitude of the equilibrium constant for an ionization reaction can be used to determine the relative strengths of acids and bases. The equilibrium constant for this reaction is the base ionization constant K b , also called the base dissociation constant:. Once again, the activity of water has a value of 1, so water does not appear in the equilibrium constant expression.
A: The acid given is phosphoric acid i. Which statement always accurately describes acids and bases a the conjugate base of a weak acid is a strong base b the conjugate acid of a weak base is a weak acid c strong acids and st? A: The compound which can donate proton is called acid, and after donating proton the anion formed…. What is conjugate acid and conjugate base? A: Ans given as follows. Standard XII Chemistry. A little amount of X when tested with blue litmus turns to red. Ka for… A: The more ka value of acid the more acidic and the conjugatebase of that strong acid is weakest…. For example: The acid-base reaction forms conjugate acid and base by the addition and removal of the proton. John W.

In it something is. Many thanks for the help in this question.